Previous | Next
China-Sri Lanka Joint Research and Demonstration Centre for Water Technology
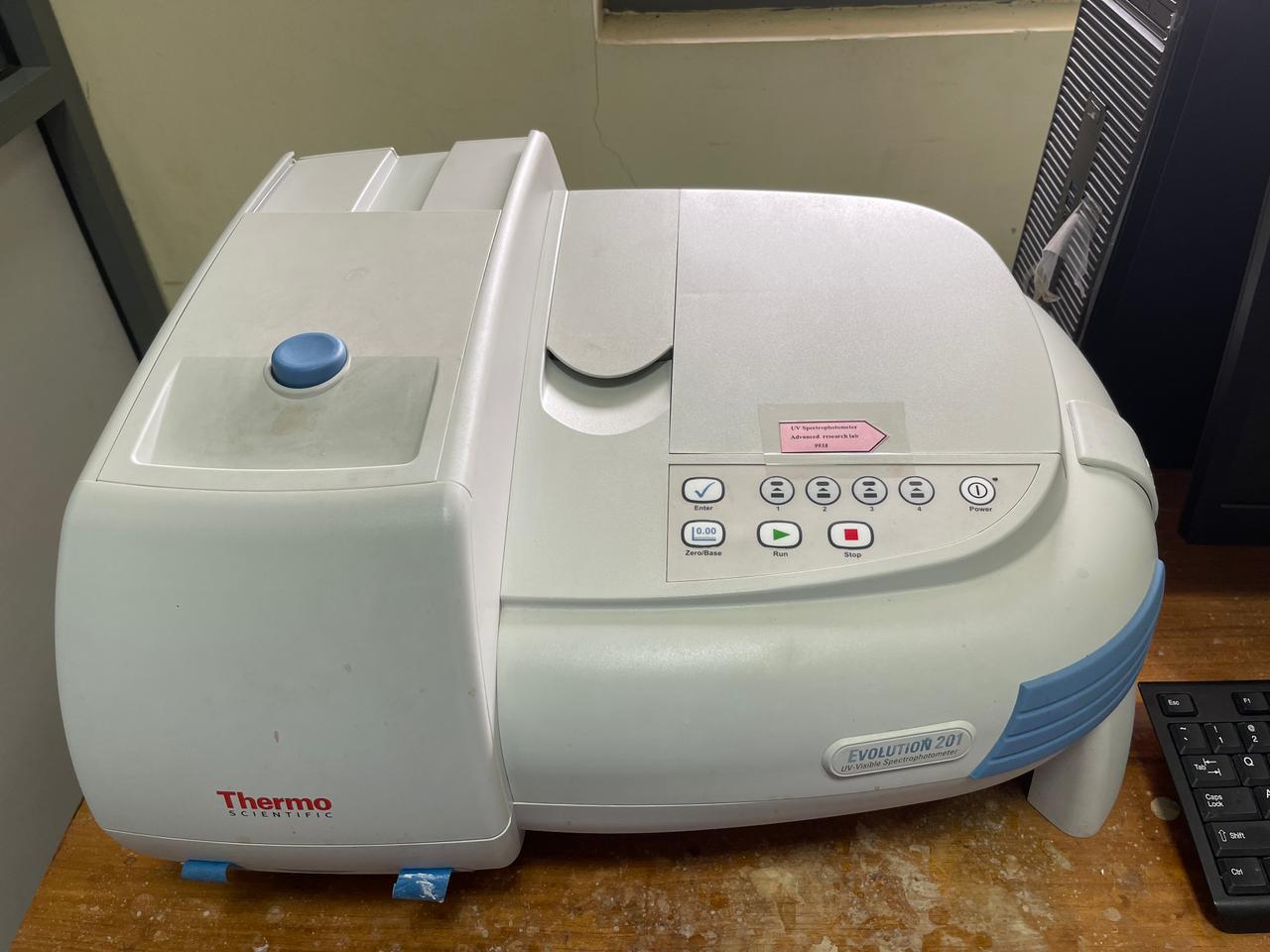
Total Phosphorus Water Quality Analyzer
China-Sri Lanka Joint Research and Demonstration Centre for Water Technology
Product Category/ Test Name (Matrix):
Water- Total Phosphorus
Sub Category:
Surface Water, Groundwater, Seawater, Drinking water sources, Domestic sewage.
Keywords:
Service Charge (Rs) - 



















.jpg)
.jpg)
































.jpg)







































.jpg)


.jpg)
.jpg)


.jpg)
1.jpg)
.jpg)
.jpg)
.jpg)






.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)






.jpg)


































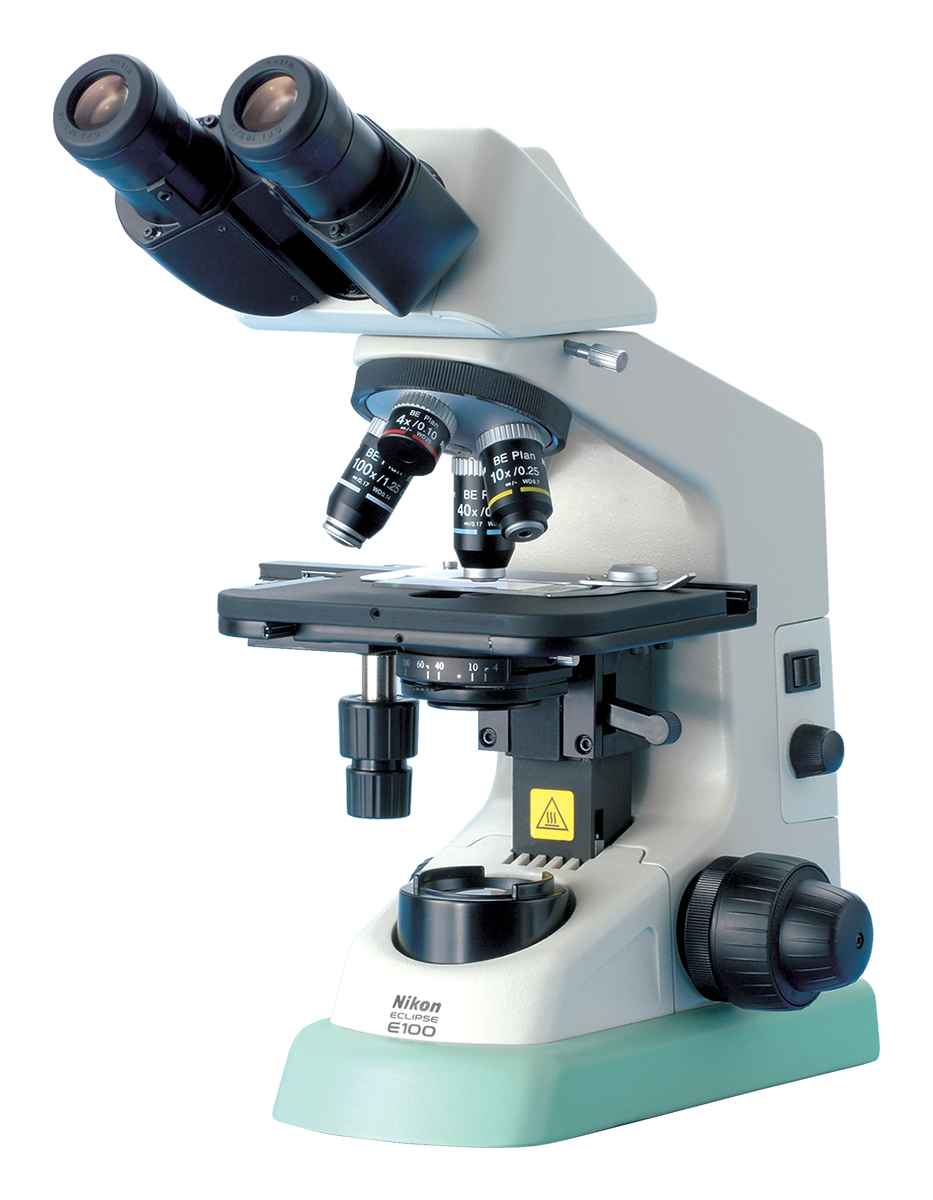

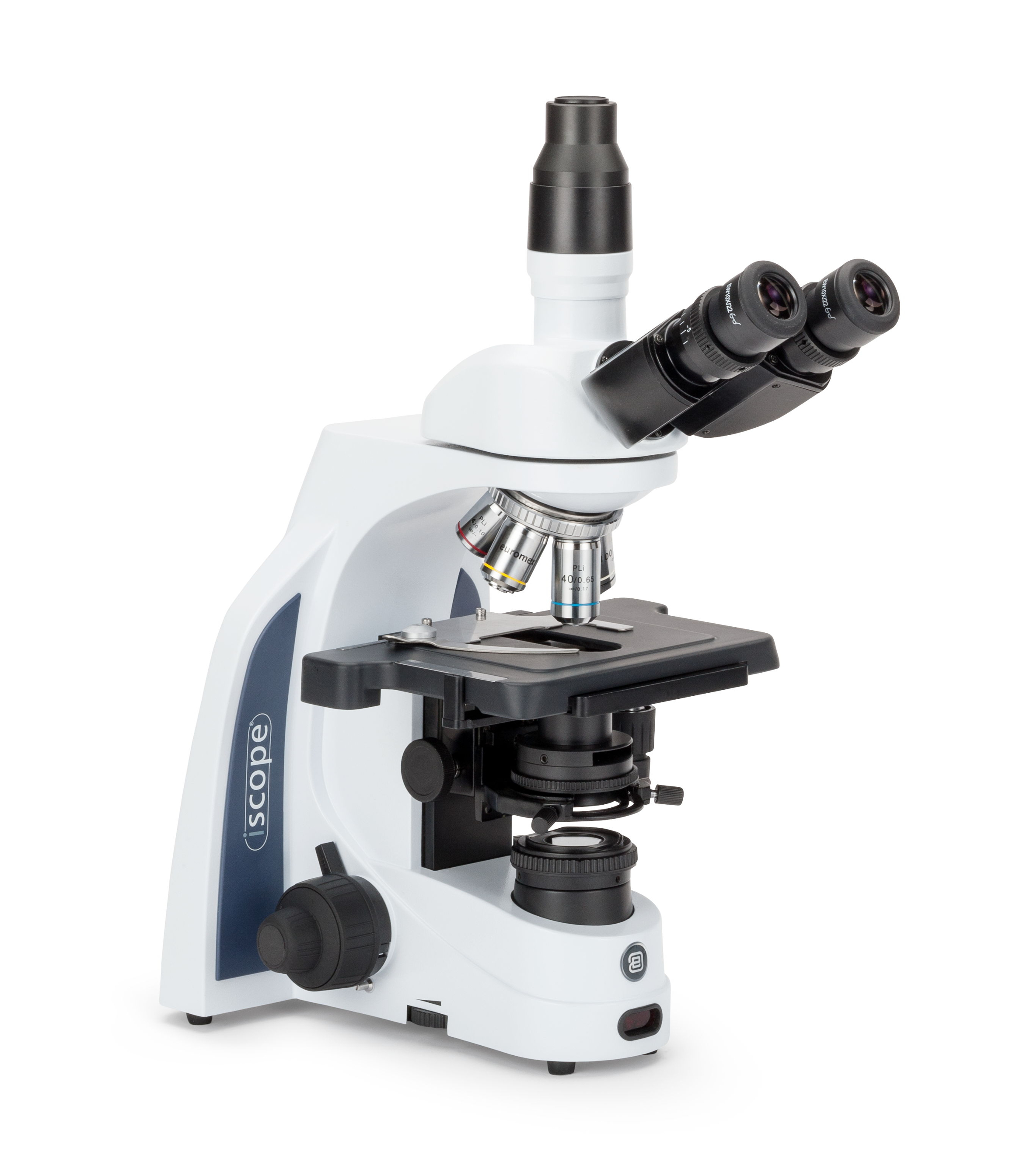
















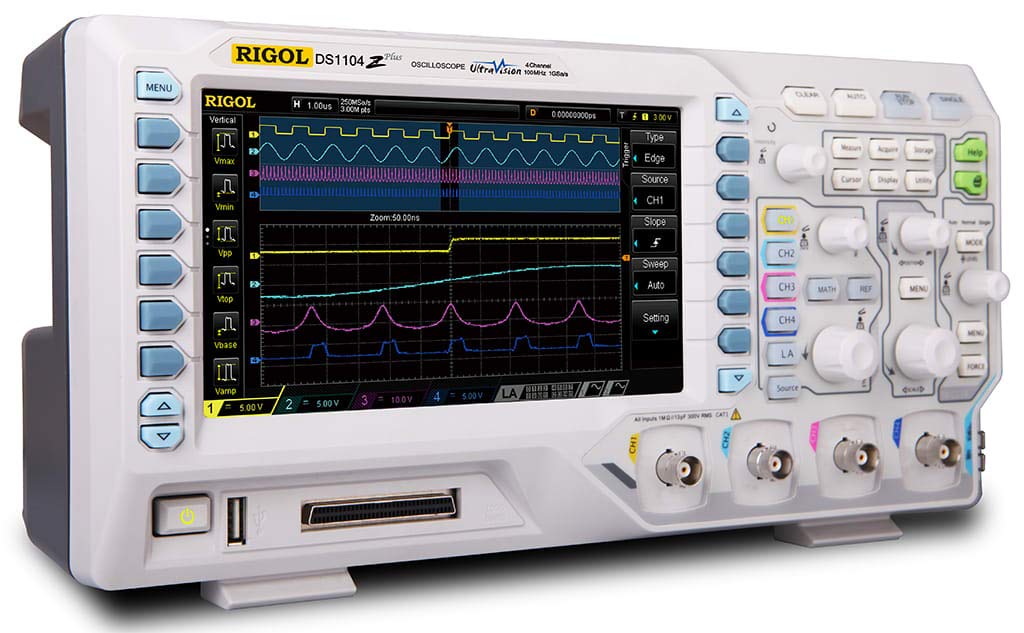



















































.jpg)
.jpg)





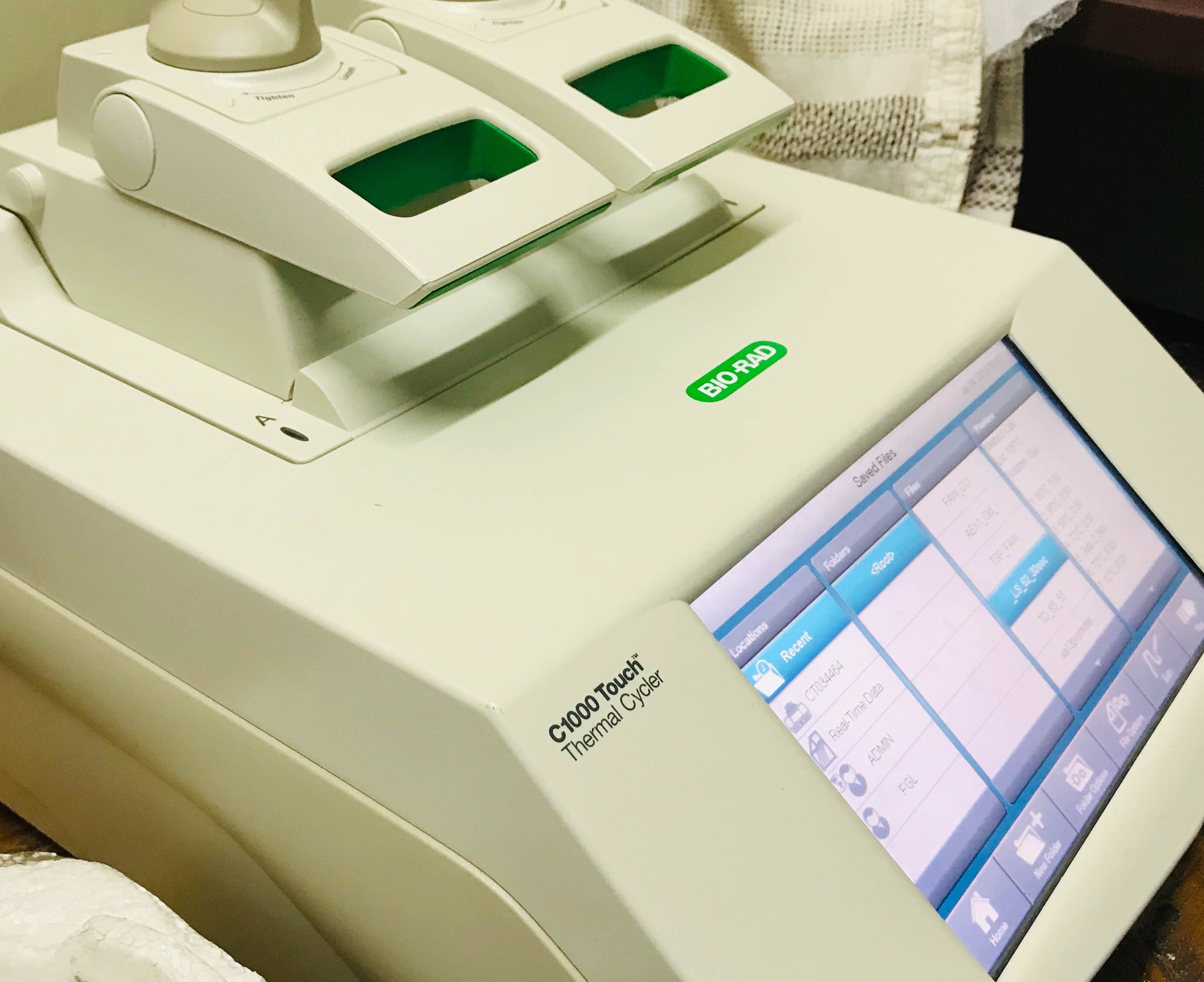





















.jpg)

























.jpg)































1.jpg)







1.jpg)





































.jpg)











.jpg)






















.jpg)










































.jpg)


















.jpg)


.jpg)




.jpg)







































































3.jpg)

























_FN_500P.jpg)

























.jpg)
.jpg)
.jpg)










1.jpg)

































.jpg)
.jpg)



